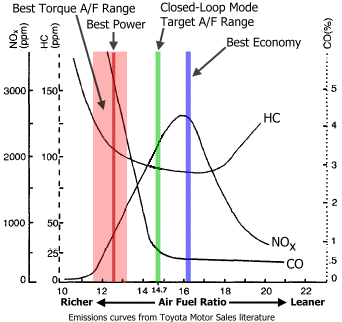
Wondering about fuel economy verus emissions
#1
Elite Member
Thread Starter
iTrader: (5)
Join Date: Mar 2007
Location: NYC
Posts: 1,970
Total Cats: 1
Let me preface this with acknowledging that most here do not care much about how they pollute. I understand that, so no need to tell me I shouldn't worry about it or other comments like that.
When going with an aftermarket ECU like MS most people lean out their cruise AFRs as much as possible to increase gas mileage, but this also increases emmisions. It's my understanding that the cleanest burn is at ~14.7 but obviously not the most fuel efficient.
What do you think has a more of a negative environmental impact, using less fuel by leaning out cruise but polluting more per mile; or keeping it at the optimal ratio for a clean burn but having to use more fuel?
edit: f-d up the title
When going with an aftermarket ECU like MS most people lean out their cruise AFRs as much as possible to increase gas mileage, but this also increases emmisions. It's my understanding that the cleanest burn is at ~14.7 but obviously not the most fuel efficient.
What do you think has a more of a negative environmental impact, using less fuel by leaning out cruise but polluting more per mile; or keeping it at the optimal ratio for a clean burn but having to use more fuel?
edit: f-d up the title
Last edited by johndoe; 06-18-2008 at 12:51 PM.
#2
Cleanest burn is not necessarily at 14.7, its just that the cat works most efficiently at 14.7. Running lean does reduce HC & CO emissions, but only up to the point of misfire. NOx emissions on the other hand increase when running leaner than 14.7.
So yeah, to summarize, if you are running leaner on a cat-equipped engine, you will reduce HC/CO emissions, but NOx will go up significantly the moment you start to deviate from 14.7. This can be offset by playing with the EGR solenoid, and such a thing can be controlled with the existing MS, its just that no one is willing to put in the time to do that.
So yeah, to summarize, if you are running leaner on a cat-equipped engine, you will reduce HC/CO emissions, but NOx will go up significantly the moment you start to deviate from 14.7. This can be offset by playing with the EGR solenoid, and such a thing can be controlled with the existing MS, its just that no one is willing to put in the time to do that.
#3
Elite Member
Thread Starter
iTrader: (5)
Join Date: Mar 2007
Location: NYC
Posts: 1,970
Total Cats: 1
That much I know (or have a vague understanding of). I'm curious more to the benefit of less emissions vs the benefit of less fuel being consumed, in the context of environmental conservation.
#6
http://www.transportation.anl.gov/software/GREET/
#7
2 Props,3 Dildos,& 1 Cat


iTrader: (8)
Join Date: Jun 2005
Location: Fake Virginia
Posts: 19,338
Total Cats: 573
Anyway, Johndoe, I think you should take something away from those graphs I posted:
You dont really gain much from minimizing NOx values. It's like a 20% penalty (increase) in NOx with virtually no change in CO or HC and hopefully a good gas savings. Plus it's only in cruise and if you get 20% better gas mileage, maybe it offsets the 20% increase in NOx?
And actually now that I'm thinking about it, isn't quite a bit of NOx production related to the heat of combustion? Perhaps if you did a light water injection you could reduce combustion temps and reduce NOx production. Hmmmmm. we need an exhaust gas analyzer.
You dont really gain much from minimizing NOx values. It's like a 20% penalty (increase) in NOx with virtually no change in CO or HC and hopefully a good gas savings. Plus it's only in cruise and if you get 20% better gas mileage, maybe it offsets the 20% increase in NOx?
And actually now that I'm thinking about it, isn't quite a bit of NOx production related to the heat of combustion? Perhaps if you did a light water injection you could reduce combustion temps and reduce NOx production. Hmmmmm. we need an exhaust gas analyzer.
#8
NOx = various oxides of nitrogen, a required precursor to smog
HC = hydrocarbons, the other ingredient to smog, also carcinogenic
CO = carbon monoxide, poisonous
CO2 = carbon dioxide, inert, byproduct of combustion
H2O = water, inert
O2 = oxygen, we breathe, necessary for life, consumed in combustion
N2 = nitrogen, inert, composes ~70% of air breathed
You need to distinguish between "engine out" emissions and "catalyst out" emissions. NOx is a result of combustion temps, the higher the temps the more NOx is created as a result of N2 + O2 <---> NOx equilibrium reaction. After the temps drop from the expansion of the gas in the power stroke, the equilibrium that was reached at high temps gets "locked in". The NOx can't auto reduce back to the N2 natural state that composes 70% of our air.
The modern 3-way catalyst has a two stage process. First it oxidizes the NO to a NO2 formula and also oxidizes the CO to CO2. This preparatory step is required to allow the NOx reduction reaction to take place on the next catalyst stage. CO oxidization is the easiest part of catalytic reduction and is usually not the most concerning factor developing an emissions approved engine.
Rich burn is preferred not just for quenching the reaction to reduce initial engine out NOx, the excess HCs from rich combustion are used by the catalyst as part of the NOx reduction reaction.
The function of EGR is also to reduce NOx production by introducing an inert gas that absorb some of the combustion temperature. This works because the piston still draws in the same mass of air, but requires less fuel (less energy, less heat) while maintaining rich burn.
Water injection can effectively achieve the same thing, but care must be taken not to wash oil from the cylinder walls causing massive increases in cooling system heat rejection and cylinder and piston deterioration due to loss of lubricant. Or dilute the oil lubrication needed for bearings and moving parts. Or cause too much reduction of combustion temp and form soot in the chamber, piston ring pack, and oil. I do not know what affect additional water would have on the catalyst; it might not be a problem since its a natural byproduct of combustion.
So that on the surface of the catalyst you get this reaction:
NOx + HC --> N2 + H2O + CO2 (+ heat)
The right proportion of HC:NOx is required to maintain a good reduction and prevent fouling of the catalyst.
The only good way to develop this is to use an engine dyno with emissions datalogging with the full engine control parameters: fuel injection timing, pressure, quantity, ignition timing, and valve actuation control, variable EGR controls, etc. Only this equipment can provide a full map for cold start, hot start, high altitude, high humidity, low humidity, low octane fuel, high octane fuel, ethanol cut fuel, a new phase of the moon, etc...
Reducing NOx is very important, I like having blue skies and fresh air. Thanks for your effort and good luck.
HC = hydrocarbons, the other ingredient to smog, also carcinogenic
CO = carbon monoxide, poisonous
CO2 = carbon dioxide, inert, byproduct of combustion
H2O = water, inert
O2 = oxygen, we breathe, necessary for life, consumed in combustion
N2 = nitrogen, inert, composes ~70% of air breathed
You need to distinguish between "engine out" emissions and "catalyst out" emissions. NOx is a result of combustion temps, the higher the temps the more NOx is created as a result of N2 + O2 <---> NOx equilibrium reaction. After the temps drop from the expansion of the gas in the power stroke, the equilibrium that was reached at high temps gets "locked in". The NOx can't auto reduce back to the N2 natural state that composes 70% of our air.
The modern 3-way catalyst has a two stage process. First it oxidizes the NO to a NO2 formula and also oxidizes the CO to CO2. This preparatory step is required to allow the NOx reduction reaction to take place on the next catalyst stage. CO oxidization is the easiest part of catalytic reduction and is usually not the most concerning factor developing an emissions approved engine.
Rich burn is preferred not just for quenching the reaction to reduce initial engine out NOx, the excess HCs from rich combustion are used by the catalyst as part of the NOx reduction reaction.
The function of EGR is also to reduce NOx production by introducing an inert gas that absorb some of the combustion temperature. This works because the piston still draws in the same mass of air, but requires less fuel (less energy, less heat) while maintaining rich burn.
Water injection can effectively achieve the same thing, but care must be taken not to wash oil from the cylinder walls causing massive increases in cooling system heat rejection and cylinder and piston deterioration due to loss of lubricant. Or dilute the oil lubrication needed for bearings and moving parts. Or cause too much reduction of combustion temp and form soot in the chamber, piston ring pack, and oil. I do not know what affect additional water would have on the catalyst; it might not be a problem since its a natural byproduct of combustion.
So that on the surface of the catalyst you get this reaction:
NOx + HC --> N2 + H2O + CO2 (+ heat)
The right proportion of HC:NOx is required to maintain a good reduction and prevent fouling of the catalyst.
The only good way to develop this is to use an engine dyno with emissions datalogging with the full engine control parameters: fuel injection timing, pressure, quantity, ignition timing, and valve actuation control, variable EGR controls, etc. Only this equipment can provide a full map for cold start, hot start, high altitude, high humidity, low humidity, low octane fuel, high octane fuel, ethanol cut fuel, a new phase of the moon, etc...
Reducing NOx is very important, I like having blue skies and fresh air. Thanks for your effort and good luck.

#9
That's some good info drewtam.
I would say it comes down to a choice between more focussed type of emissions, such os NOx and more CO2. More smog and lung problems or more global warming. It's the same problem presented with diesel engines. Europe creates far less greehouse gas emissions than we do, because of heavy use of diesel, but they also have higher rates of asthma. We on the other hand have pretty "clean" air, but put out a lot more green house gas emissions.
With the balance towards greenhouse gasses in this country I would tend toward using less fuel and increasing NOx output just for the "balanced" approach. Of course running water/alcohol injection, reducing NOx, keeping the mileage gains AND being able to run more boost and make more power is the great ideal. Hell, I may have just convinced myself to pick up a Devils Own...
I would say it comes down to a choice between more focussed type of emissions, such os NOx and more CO2. More smog and lung problems or more global warming. It's the same problem presented with diesel engines. Europe creates far less greehouse gas emissions than we do, because of heavy use of diesel, but they also have higher rates of asthma. We on the other hand have pretty "clean" air, but put out a lot more green house gas emissions.
With the balance towards greenhouse gasses in this country I would tend toward using less fuel and increasing NOx output just for the "balanced" approach. Of course running water/alcohol injection, reducing NOx, keeping the mileage gains AND being able to run more boost and make more power is the great ideal. Hell, I may have just convinced myself to pick up a Devils Own...
Thread
Thread Starter
Forum
Replies
Last Post