The AI-generated cat pictures thread
That has been the main problem so far with running pure oxygen. We have not found a way to store it. imagine an accident where the crazy high pressures scuba tank gets damaged.. you have either a bomb or a missile.
The ordinary combustion engine is garbage. If you want to talk about pure hydrogen vehicles a better method is using a fuel cell to get electricity out to run an electric motor. You'll end up with total vehicle efficiency in the 50%.
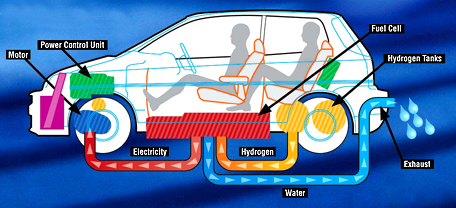
The ordinary combustion engine is garbage. If you want to talk about pure hydrogen vehicles a better method is using a fuel cell to get electricity out to run an electric motor. You'll end up with total vehicle efficiency in the 50%.

It is also worth noting that oxygen isn't flammable, nor is it combustible. A storage method that secures a high pressure LOX tank to the frame of a car is *relatively* secure so long as the high pressure tank is not allowed to gain any momentum if it is punctured. Any oxygen that is released from a rupture of such a tank will not cause any object to catch fire unless that object is already alight/burning/embered.
I'm interested in seeing the MPG difference for a pickup truck full of HHO generators.
It is also worth noting that oxygen isn't flammable, nor is it combustible. A storage method that secures a high pressure LOX tank to the frame of a car is *relatively* secure so long as the high pressure tank is not allowed to gain any momentum if it is punctured. Any oxygen that is released from a rupture of such a tank will not cause any object to catch fire unless that object is already alight/burning/embered.
It is also worth noting that oxygen isn't flammable, nor is it combustible. A storage method that secures a high pressure LOX tank to the frame of a car is *relatively* secure so long as the high pressure tank is not allowed to gain any momentum if it is punctured. Any oxygen that is released from a rupture of such a tank will not cause any object to catch fire unless that object is already alight/burning/embered.
 .
.Oxygen it self is not flammable however you should know that gasoline by it self is not flammable either. The bigger problem here is the hydrogen though. A leaking hydrogen tank is a giant problem.
Storage method that secures high pressure LOX tanks to a car? A broken 3000 psi tank can output 3000 psi of thrust. Next problem is you will have multiple tanks next to eachother. That also includes the hydrogen tanks... in pure form. Hydrogen does not ignite, it explodes.
Since we're talking about thrust heres a pic of a jet powered train.

Originally Posted by fooger03;1019219
It is also worth noting that oxygen isn't flammable, nor is it combustible. A storage method that secures a high pressure LOX tank to the frame of a car is *relatively* secure so long as the high pressure tank is not allowed to gain any momentum if it is punctured. Any oxygen that is released from a rupture of such a tank will not cause any object to catch fire unless that object is already alight/burning/embered.
[yt
It is also worth noting that oxygen isn't flammable, nor is it combustible. A storage method that secures a high pressure LOX tank to the frame of a car is *relatively* secure so long as the high pressure tank is not allowed to gain any momentum if it is punctured. Any oxygen that is released from a rupture of such a tank will not cause any object to catch fire unless that object is already alight/burning/embered.
[yt
WuG5WTId-IY[/yt]
per my 4 years of AirForce Aircraft Maintenance.
all our GOX or LOX carts have very specific step by step tasks

Boost Pope


iTrader: (8)
Join Date: Sep 2005
Location: Chicago. (The less-murder part.)
Posts: 33,076
Total Cats: 6,628
Oxygen is an oxidizer.
Everything else (mostly) is a fuel.
Fuel cannot burn without an oxidizer to react with. An oxidizer, by itself, cannot burn. However, it is required in order for other things to burn.
The more oxygen is present, the more rapidly / violently other things will burn.
If you light a match and place it inside a large jar full of ordinary air, it will burn like, well... like a match. Air is about 21% oxygen.
If you suddenly remove all of the oxygen from the jar and replace it with some other gas (helium, nitrogen, hell, even a mixture of gasoline vapor and hydrogen), the match will go out.
If you instead add more and more oxygen (above 21%), the match will burn brighter and faster. If you add 100% pure oxygen at a high pressure, it will burn almost explosively.
But they key point is that it's not the oxygen that's burning, it's the match. The oxygen is merely enabling the match to burn.
This is what we are seeing in the "how to light a grill" video. The coals are burning, and the oxygen is enabling them to burn.
Consider an oxy/acetylene torch:
You start with just acetylene. It burns, but not terribly well given the relatively low amount of oxygen it has to work with (the free oxygen in the ambient air.)
Next, you start turning on the oxygen. As you add oxygen to the mixture, the acetylene is able to burn much more rapidly and much hotter.
If you then turn off the acetylene, the torch will go out entirely. This is because you've taken away the fuel source, and the oxygen, by itself, cannot burn.
This is also why nitrous oxide works. During the combustion process, it rapidly decomposes into nitrogen and oxygen. This additional oxygen, above and beyond the amount of oxygen which would typically be present in the chamber, permits more gasoline to be completely burned than would otherwise be possible.
Everything else (mostly) is a fuel.
Fuel cannot burn without an oxidizer to react with. An oxidizer, by itself, cannot burn. However, it is required in order for other things to burn.
The more oxygen is present, the more rapidly / violently other things will burn.
If you light a match and place it inside a large jar full of ordinary air, it will burn like, well... like a match. Air is about 21% oxygen.
If you suddenly remove all of the oxygen from the jar and replace it with some other gas (helium, nitrogen, hell, even a mixture of gasoline vapor and hydrogen), the match will go out.
If you instead add more and more oxygen (above 21%), the match will burn brighter and faster. If you add 100% pure oxygen at a high pressure, it will burn almost explosively.
But they key point is that it's not the oxygen that's burning, it's the match. The oxygen is merely enabling the match to burn.
This is what we are seeing in the "how to light a grill" video. The coals are burning, and the oxygen is enabling them to burn.
Consider an oxy/acetylene torch:
You start with just acetylene. It burns, but not terribly well given the relatively low amount of oxygen it has to work with (the free oxygen in the ambient air.)
Next, you start turning on the oxygen. As you add oxygen to the mixture, the acetylene is able to burn much more rapidly and much hotter.
If you then turn off the acetylene, the torch will go out entirely. This is because you've taken away the fuel source, and the oxygen, by itself, cannot burn.
This is also why nitrous oxide works. During the combustion process, it rapidly decomposes into nitrogen and oxygen. This additional oxygen, above and beyond the amount of oxygen which would typically be present in the chamber, permits more gasoline to be completely burned than would otherwise be possible.
Last edited by Joe Perez; 06-07-2013 at 12:18 AM.
Even better launch (not the Rx8)
My poor attempt at photoshopping this badass manifold:
https://www.miataturbo.net/fabulous-...4/#post1019311

https://www.miataturbo.net/fabulous-...4/#post1019311